| Feature 1 | 3D True Color |
|---|---|
| Feature 2 | Calibration-Free |
| Feature 3 | Multilingual System (15 Languages) |
| Feature 4 | Humanized Scanner Tip |
| Feature 5 | Fashion and Light Handpiece |
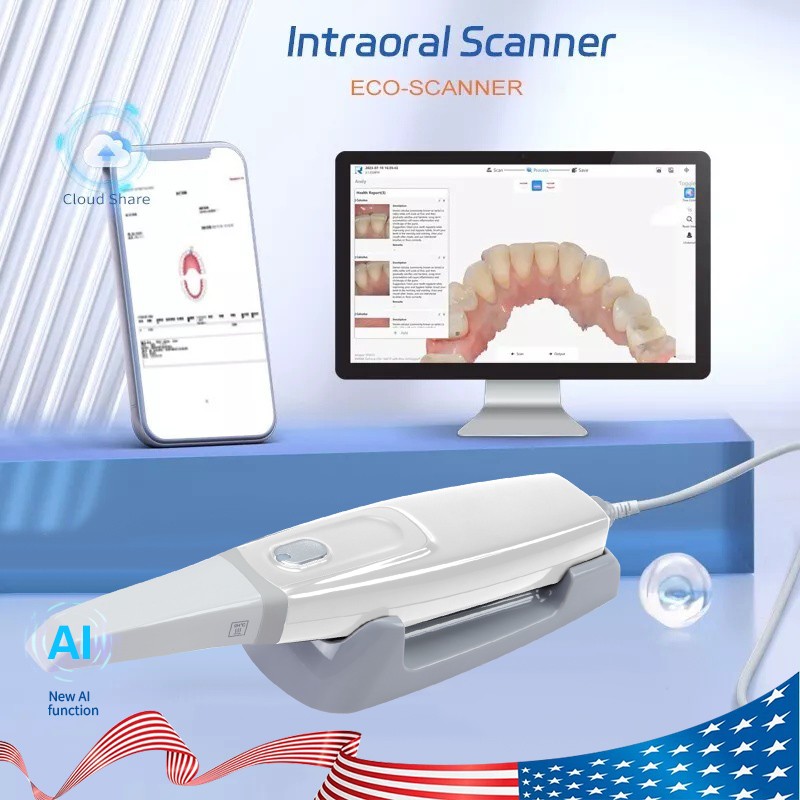
| Feature 6 | Cloud Sharing |
| Feature 7 | Complete Ecosystem |
| Feature 8 | Scan Body Matching |
| Product | Acquisition Scanner |
| FPS | 15 |
| Accuracy | Single crown≤10um, single arch≤15um, full arch≤20um |
| Scanner Weight | 210g |
| Export File | STL, PLY, OBJ |
| Operating Time | >30000hrs |
| Imaging Mode | Synchronous 3D Video |
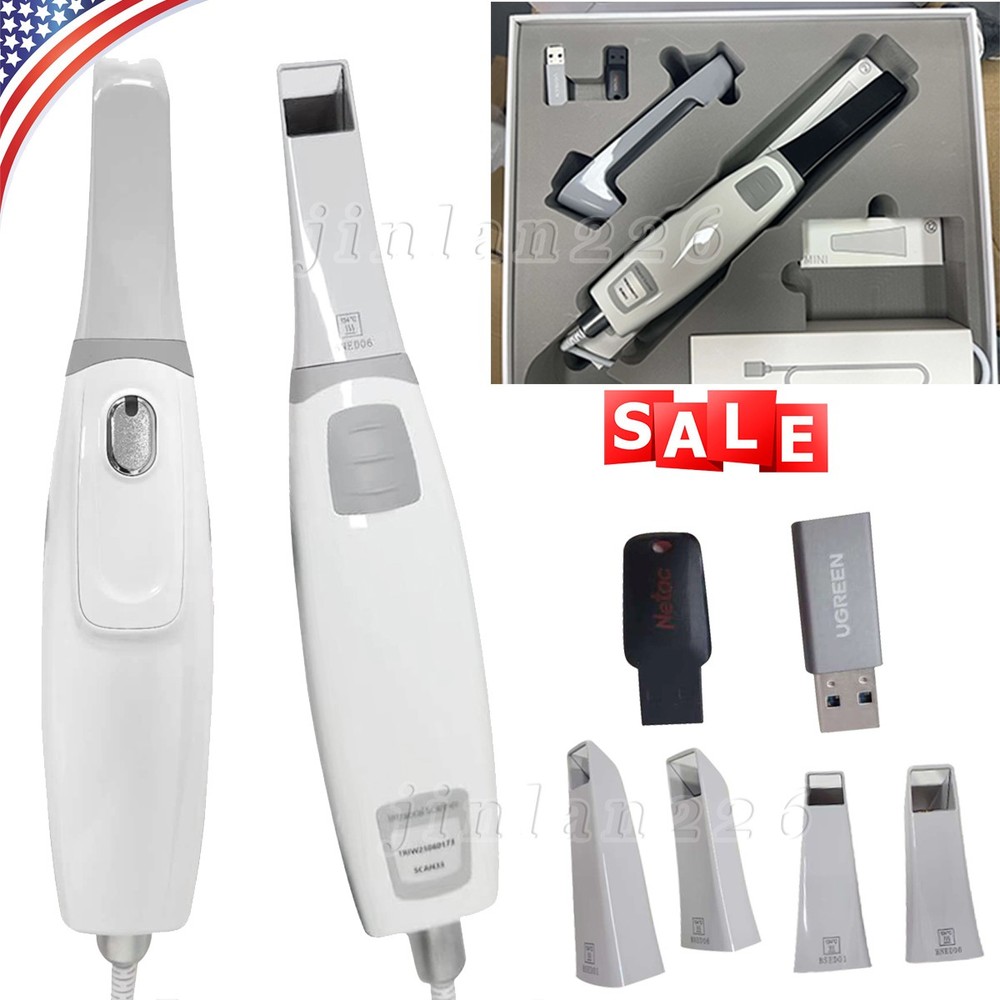
| Model | Scan 33 |
| Country of Origin | China |
| Scanner Tip | 4 Tips (3 Normal Tip + 1 Smaller Tips) |
| Attention | Not include Scanner Cart and Computer |
| Tip Autoclavable | Up to 100 Times Autoclavable 121° 16mins /134° 4mins |
| Remote Control Function | YES |
| Scanner Tips | Single Arch:30s, Full arch w/ Occlusion:90s |
| Scanning Distance | 0-15mm |
| Image Capture Technology | Optical Continuous Video Collection |
| Scanner Light Source | LED |
| FOV | 13X13mm |
| Scanner Size | 240x49.5x30.5mm |
| Oral Health Report 1 | Generate Report Automatically |
| Oral Health Report 3 | Support Printing Report |
| Oral Health Report 2 | Real-Time Review |
| FDA 510(k)number | K181691 |
| Sale | Factory Direct Sale |
| Nose cone | 1 |
| 510(K) Number For Scaler | K163414 |
| Custom Bundle | No |
| Subsequent Product Code for dental handpiece | EGS |
| Modified Item | No |
| 510(K) Number For Dental Handpiece | K181691 |
| Classification Product Code for dental handpiece | EFB |
| 510(K) Number For Curing Light | K192233 |
| Regulation Number for dental handpiece | 872.4200 |
| Brand | Unbranded |
| Intended Use/Discipline | Dental Laboratory, Dentistry |
Check the listing for details. Dental Digital Impression Intraoral 3D Scanner Scanning Software CAD/CAM Process. Condition: New, Made in China. Listed at 2599.00 USD. And it will continue to be updated! The sale of this item may be subject to regulation by the U.S. Food and Drug Administration and state and local regulatory agencies. FDA for Curing Light: 510(K) Number: K192233, Regulation Number: 21 CFR 872.6070, Product Code: EBZ.