| Approved | FDA |
|---|---|
| FDA for Curing Light | 510(K) Number: K192233 |
| FDA for Handpiece | 510(K) Number: K181691 |
| FDA for Scaler | 510(K) Number: K163414 |
| Handpiece Type | Low Speed |
| Model | JR |
| Brand | Unbranded |
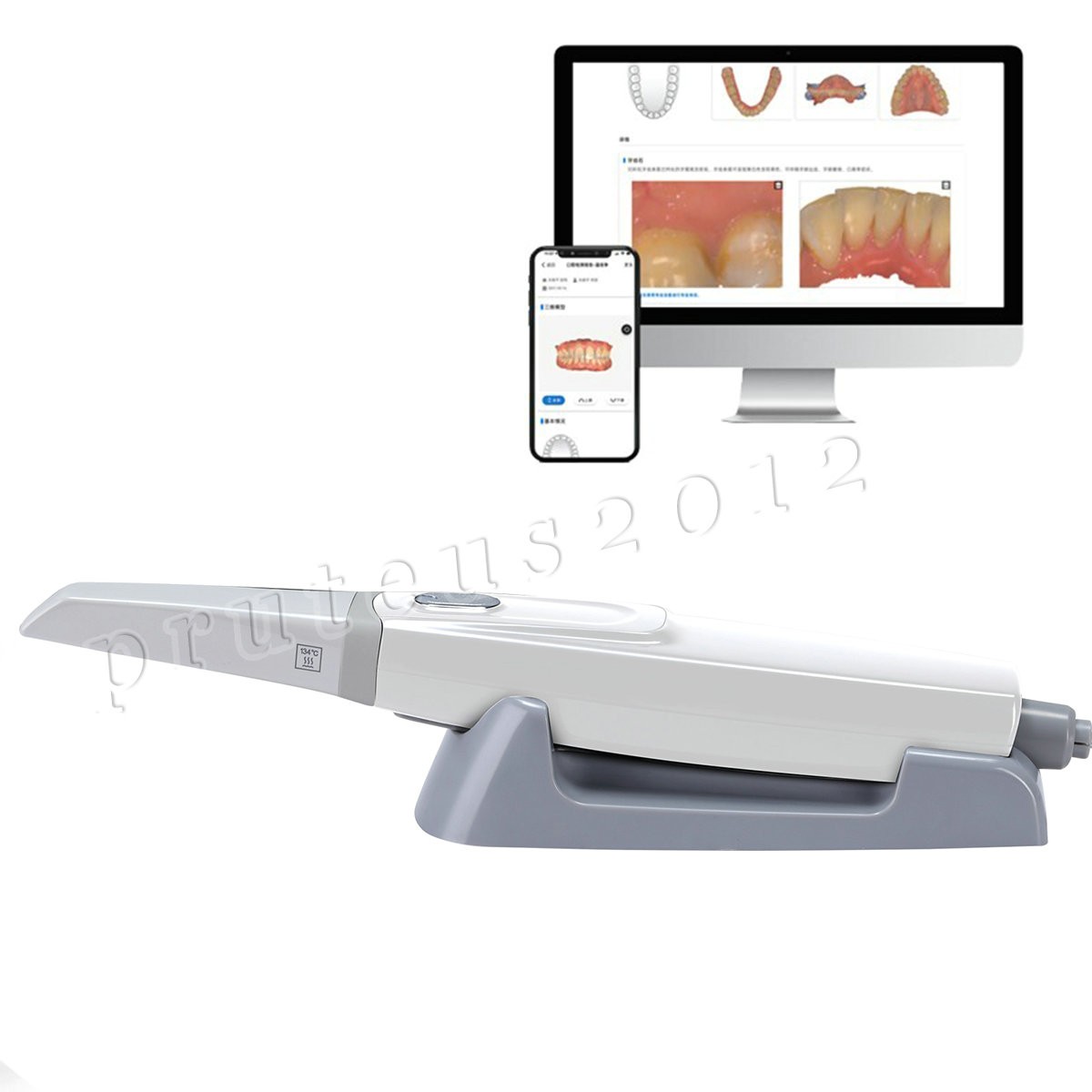
Check the listing for details. Dental Intraoral Scanner 3D Real-time Display With Scanning Software. Condition: New. Listed at 2599.00 USD. And it will continue to be updated! The sale of this item may be subject to regulation by the U.S. Food and Drug Administration and state and local regulatory agencies. City, State: Beijing, China. FDA for Handpiece: 510(K) Number: K181691, Regulation Number: 872.4200, Classification Product Code: EFB, Subsequent Product Code: EGS.


![Shining3D [ Aoralscan 3 ] Dental Intra-oral 3D Scanner with Scanning Software](/image/iqQAAOSwg1thYhsk/shining3d-aoralscan-3-dental-intra-oral-3d-scanner-with-scanning-software.jpg)