| Item Package Quantity | 1 |
|---|---|
| Brand | EKO |
| Fda Device Classification | Ii |
| Temperature Rating | Ambient: 15 To 25 C (59 To 77 F) |
| Fda Indication Of Use | Otc |
| Part Number | Cor501-Parent |
| Fda Instructions For Use Type | Consumer |
| Number Of Lithium Ion Cells | 1 |
| Color | Matte Black |
| Battery Weight | 20.0 |
| Number Of Boxes | 1 |
| Model Number | Cor501-Blk/Blk |
| Fda Label Type | Consumer |
| Batteries Required? | Yes |
| Unit Count | 1.0 |
| Batteries Included? | Yes |
| Fda Premarket Approval Number 510K | K233609 |
| EAN | 0850010298290 |
| ISBN | 0850010298290 |
| Package Dimensions LxWxH | 18.75x6.75x2.5 Inches |
| Weight | 2.2 Pounds |
| UPC | 850010298290 |
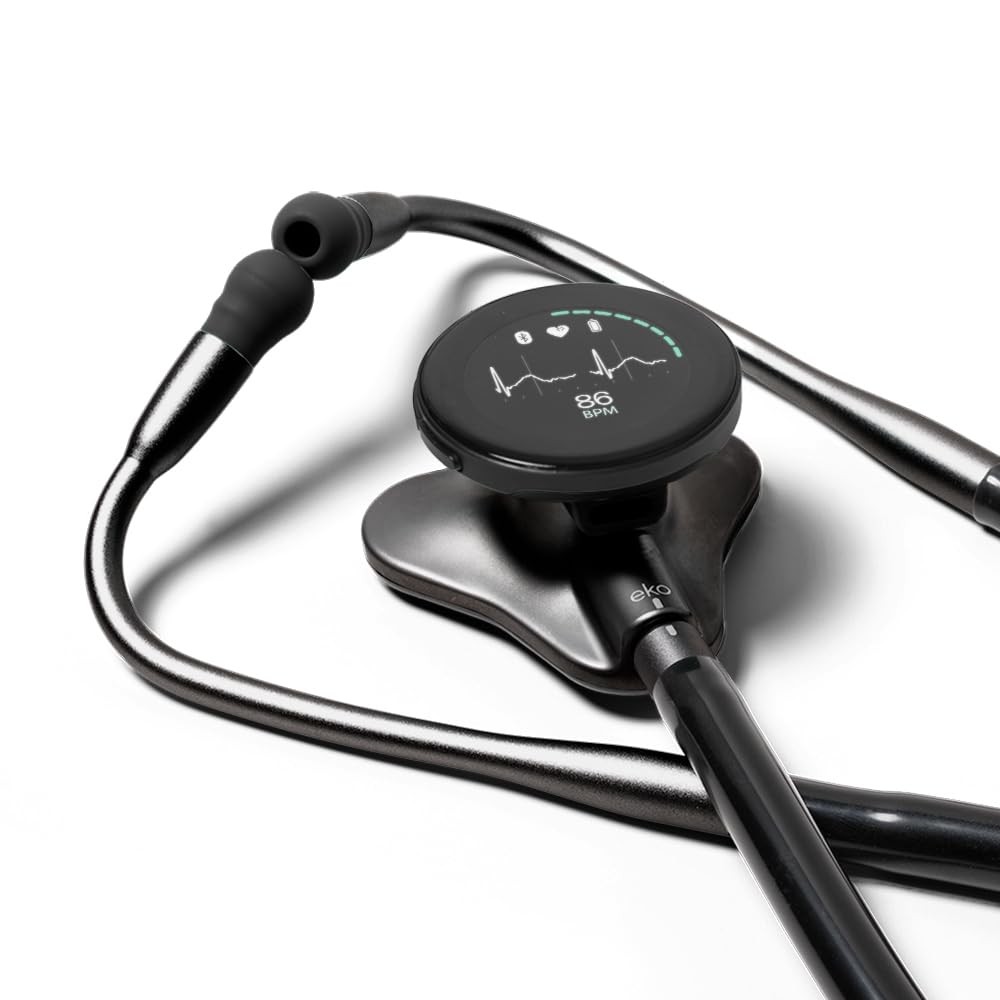
Check the listing for details. Eko CORE 500™ Digital Stethoscope — 40x Amplification, Active Noise Cancellat.... Color: Matte Black, Condition: New. Listed at 718.04 USD. Even without Eko+, you can enjoy basic features of the app. Eko AI capabilities are supported by separate Eko AI products. A software subscription may be required to access select Eko App features.











