| Fda Premarket Approval Number Pma | K173140 |
|---|---|
| Model Name | Vgm09 |
| Fda Device Classification | Ii |
| Fda Indication Of Use | Otc |
| Operating Time | 5.0 |
| Part Number | Vgm09 |
| Fda Instructions For Use Type | Consumer |
| Model Number | Vgm09 |
| Fda Label Type | Consumer |
| Batteries Required? | Yes |
| Fda Premarket Approval Number 510K | K173140 |
| EAN | 0763351997763 |
| ISBN | 0763351997763 |
| Package Dimensions LxWxH | 4.92x4.17x2.24 Inches |
| Weight | 0.51 Pounds |
| UPC | 763351997763 |
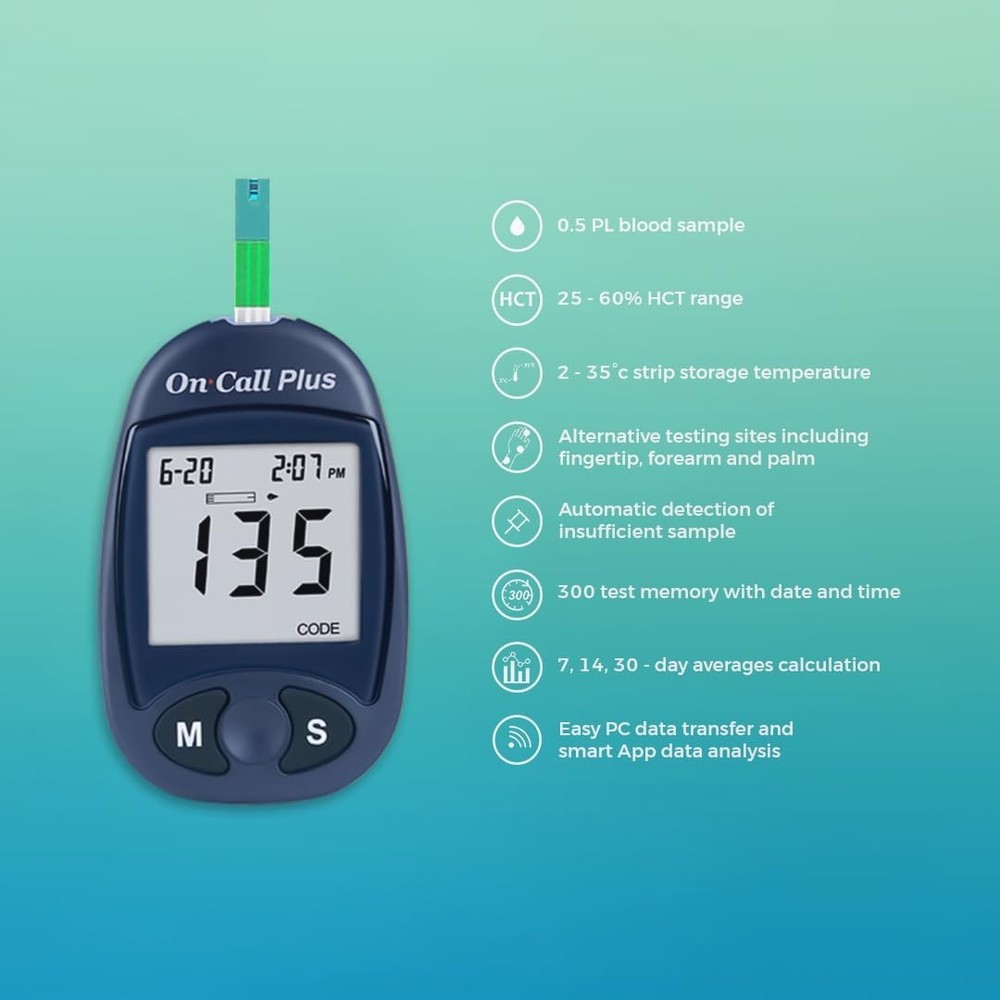
Check the listing for details. IMDK Blood Glucose Monitor Kit, 𝗖𝗹𝗶𝗻𝗶𝗰𝗮𝗹𝗹𝘆 𝗔𝗰𝗰𝘂𝗿𝗮𝘁𝗲 Blood S.... Condition: New. Listed at 46.81 USD. It has certifications from FSA/HSA CE, GMP, ISO 13485:2013 and ISO 15197:2013. Trust your results and gain a clearer picture of your blood sugar with IMDK.